Convenience store retailers are still concerned over the FDA’s involvement in smokeless tobacco, but many expressed optimism that the category can continue to thrive. By Howard Riell, Associate Editor. While taxes and regulations haven’t impacted sales of smokeless tobacco products as much as some had predicted, it remains up to retailers to continue aggressively marketing…
FDA Seeks Comments on Draft Guidance
FDA has posted a draft guidance to help persons submitting applications for new tobacco products understand the premarket tobacco product application process. The draft guidance addresses questions such as: • Who may submit a new tobacco product application • When to submit a new tobacco product application • What information the FD&C Act requires you…
Tobacco Companies Fight Warning Labels
Hearing underway in U.S. Federal District Court on cigarette health warnings. Yesterday, Sept. 21, a hearing took place before U.S. Federal District Court Judge Richard Leon on a lawsuit filed by five tobacco manufacturers seeking a preliminary injunction against the implementation of the FDA’s new text and graphic picture warning labels for cigarette packages and…
FDA Seeks Comments On Draft Guidance
FDA Center for Tobacco Products posted two Draft Guidance documents and an Advance Notice of Proposed Rule-Making, and encourages retailers to review the documents and submit comments. • Draft Guidance for Industry and FDA Staff Demonstrating the Substantial Equivalence of a New Tobacco Product: Responses to Frequently Asked Questions. This draft guidance provides information in…
FDA Center for Tobacco Products Invites Retailer Comments
Retailers with comments on tobacco regulations, risk, etc., have a chance to present their views to the FDA’s Center for Tobacco (CTP) products: 1. Public Workshop on Modified Risk Tobacco Product Applications (MRTP) This workshop <http://www.fda.gov/TobaccoProducts/NewsEvents/ucm268472.htm> will provide an opportunity to discuss issues pertinent to the scientific evaluation of MRTP applications. Aug. 25 – 26,…
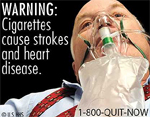
Mississippi Retailer Challenges Tobacco Warning Labels
A Mississippi tobacco retailer is questioning the U.S. Food and Drug Administration’s plan that will require tobacco makers to include graphic health warnings and graphic images on cigarette packaging and advertisements, “It’s another form of Big Daddy trying to tell people what to do,” said Frank Williams of Gulfport, who owns three Smokey’s Discount Tobacco…
The National Advisory Group: Building Profits and Relationships
Only one more week to capitalize on early bird registration for the 2011 NAG Ideas Conference in Savannah, Ga. There is more pressure than ever on convenience store chains to not only exceed customers’ expectations, but to stay ahead of the competition, navigate legislative issues and still be good corporate citizens in the communities they…
Assessment On Impact of Menthol Cigarettes Expected in July
The Family Smoking Prevention and Tobacco Control Act requires the FDA’s TPSAC to submit a report and recommendation on the impact of menthol cigarettes on public health. The U.S. Food and Drug Administration (FDA) plans to submit its draft independent review of the science related to the impact of menthol in cigarettes on public health…
Strong Demand for Cigars
Flavored and single-serve cigars represent a real opportunity for convenience stores to boost tobacco sales. By Howard Riell, Associate Editor. With flavored cigarettes no longer available in the U.S., flavored cigars are in line for sharp sales gains. But with FDA having declared last year that it’s got the cigar industry in its sights, retailers…
FDA’s CTP Website Features New Content
FDA seeks to educate retailers with new quizzes and updated information on tobacco regulations, including in Spanish. The FDA’s Center for Tobacco Products’ (CTP) has added new features to the retailer section of its Website. FDA hopes these new updates and quizzes will help educate retailers about relevant tobacco regulations and increase voluntary compliance with…